 Lactate Plus Meter
Lactate Plus Meter
Blood Lactate Meter for Endurance Training and Conditioning
Purchase here
The Test
Lactate Plus measures blood lactate (muscle performance/perfusion indicator) very quickly on a tiny drop of blood. This results in a virtually painless, fast, and accurate lactate testing. Lactate is the premier indicator of aerobic conditioning for athletes and a key predictor of competitive performance.Less Pain
Lactate tests on other meters are painful and therefore difficult to incorporate into routine training. They need 5 to 25 microliters of blood and require a wide-gauge lance to pierce the skin deeply enough to obtain a large sample. Lactate Plus requires only 0.7 microliters of blood. This allows for the use of the thinnest, most shallow, and least painful lancing devices available, which increases the athlete’s tolerance for testing.Fastest Analysis Time
The older technology used by other lactate meters can require as much as 60 seconds for test results. Lactate Plus takes only 13 seconds. This speed allows the ability to establish a rapid metabolic assessment and possible intervention. 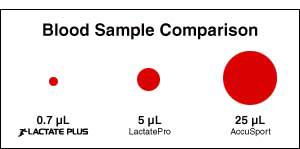 Very Accurate
Very Accurate
Lactate Plus is made by Nova Biomedical, the world leader in hospital-based, blood lactate analyzers. More hospitals worldwide use Nova for lactate measurements than any other brand. The same high standards for accuracy and reliability that are required for hospital use have been incorporated into the Lactate Plus meter.
Eliminates Calibration Strips or Calibration Codes
All other lactate strips require a unique calibration code to be entered into the meter before a strip is used. This code can be entered manually or by inserting a code strip, which comes with each vial of strips. These codes are extremely important because they can change the lactate result by up to 30%, depending on the manufacturing lot of the strip. Failing to use a code or using the wrong code can result in very inaccurate lactate results. Nova strips do not need calibration codes or strips, eliminating both a time-consuming step as well as a source of real error.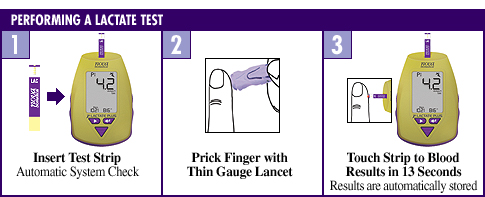 Low Cost
Low Cost
Lactate Plus meters and strips are priced less than most other similar devices.
Visit the Nova Lactate Plus store and purchase here
Nova Biomedical / 200 Prospect Street / Waltham, MA 02453-3465 / 781-894-0800
FOR PROFESSIONAL USE ONLY
Precaution: The Lactate Plus System is not FDA cleared for performing lactate measurements on persons for medical intervention. It is intended for sports training and conditioning only.
Certifications and Compliance: FDA Quality System Regulations and EN ISO 13485:2016, Complies to IVDD Tested according to: EN 61010-1:2010, EN 61010-2-101:2015, EN 60825-1/A1:2014, UL, CSA Standards FDA Labeling:For in-vitro diagnostic use. Patented Technology. Patents 6,287,451 / 6,837,976 / EP 1 212 609 / CA 2,375,092 / EP 1497 449 / JP 405 0078 Specifications subject to change without notice.